 |
John J. Bauer, M.D.
www.flinturology.com Urology Services, Inc.
|
Artifical Urinary Sphincter (AUS-800)
Surgery Details
 |
John J. Bauer, M.D.
www.flinturology.com Urology Services, Inc.
|
Artifical Urinary Sphincter (AUS-800)
Surgery Details
Contents
General information
Pre-operative instructions
Risks and Complications
Detailed Surgery Description
Family waiting instructions
Post-operative instructions
Printing tip: If you want to print only one portion of this entire document, you should be able to do this depending on your software. To print a selection, highlight the section you want to print using your mouse, then click on print, and then in the print menu, choose "selection."
Terminology tip: If you come across words you don't understand, look them up in the On-Line Medical Dictionary.
This procedure is performed to correct stress urinary incontinence in men after prostate surgery for benign tumors (BPH) and prostate cancer. This procedure places a silicone cuff prosthesis around the urethra, a small pump in the scrotum and a reservoir in the abdomen to prevent leakage of urine. (See figure below.)
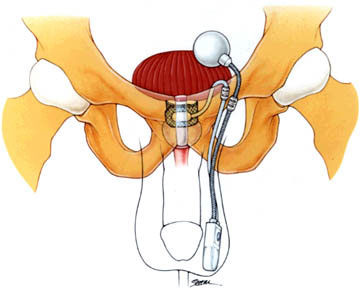
For video presentations on the urinary sphincter, you can watch the videos available at these links:
Your pre-operative appointments
Before your surgery, you will be seen by the physician and the anesthesiologist, and when applicable, there is a pre-admission appointment with the hospital. Click here to read more details about these appointments, referred to as the Pre-Operative Work-Up.
Change In Health Status
Notify your surgeon if you experience any significant change in your health status: develop a cold, influenza, a bladder infection, diarrhea, or other infection, before your surgery.
Pre-Operative Medication Instructions
Unless specifically instructed otherwise by your surgeon or anesthesiologist, please observe the following guidelines for taking your medicines before surgery:
As injury to the bowel is unlikely in this procedure, you will have the simplest form of a bowel preparation, described below.
Pre-Operative Diet Instructions
Unless specifically instructed otherwise by your surgeon or anesthesiologist, patients of all ages must observe the following diet restrictions before surgery:
Patients undergoing operative or diagnostic procedures involving sedation are required to refrain from eating, drinking or taking anything by mouth for a stated period prior to their surgery or procedure. The reason for this is to prevent complications caused by nausea or vomiting while you are unconscious. Should you vomit while in the unconscious state, the risk exists that the vomit may enter into your lungs causing serious complications such as pneumonia. These complications may result in an extension of your hospitalization following your surgical procedure. It is for this reason patients are often instructed to have nothing by mouth after midnight the night prior to your operation unless otherwise instructed by an anesthetist.
Pre-Operative Cleaning Instructions (bathing and showering instructions)
Pre-operative showers are to be taken the night before and the morning of surgery just prior to your arrival. All adults are required to take a shower using either a Betadine or Hibiclens Surgical Scrub antibacterial soap. The reason is to remove as much bacteria from your skin as possible prior to your surgery. If you are allergic to these products please notify your physician or nurse. Perform your shower as follows:
On The Day Of Surgery
The anesthesiologist will discuss with you the anesthetic most appropriate for your medical condition and procedure prior to surgery.
After your surgery you must be escorted/driven home by a responsible adult. You may take a taxi car or shuttle if accompanied by a responsible adult who can stay with you after the driver departs.
Time To Arrive For Your Surgery
During your Pre-Admission Interview, our Registered Nurse will provide you with the correct time to arrive for check-in prior to your surgery.
ARRIVAL TIME:
WHERE TO ARRIVE:
The risks and complications for this surgery are described in the "Counseling and Pre-Op Note" that you will need to sign before the surgery. The main content of that note is listed below.
Indications:
Patient is a male with stress urinary incontinence from previous prostate surgery for benign and malignant conditions or has an incompetent sphincter secondary to neurological disease. Urodynamic studies have confirmed the presence of Type III stress urinary incontinence with a low valsalva leak point pressure.
Alternatives:
Alternatives include watchful waiting, Kegel exercises, behavioral/biofeedback/electrical stimulation therapy, injection of urethral bulking agents and male sling.
Risks/Complications:
The risks and complications of the procedure where extensively discussed with the patient. The general risks of this procedure include, but are not limited to bleeding, transfusion, infection, wound infection/dehiscence, pain, scaring of tissues, failure of the procedure, potential injury to other surrounding structures, deep venous thrombosis, pulmonary embolus, myocardial infarction, heart failure, stroke, death or a long-term stay in the Intensive Care Unit (ICU). Additionally, mentioned were the possible serious complications of the anesthesia to include cracked teeth, airway damage, aspiration, pneumonia, spinal head-ache, nerve damage, spinal canal bleeding and malignant hyperthermia. Your anesthesiologist will discuss the risks and complications in more depth separately. Additional procedures may be necessary.
The specific risks of this procedure include, but are not limited to: persistent incontinence, late failure of the AUS by pump/reservoir/cuff failure and fluid leakage with resultant recurrent incontinence and require partial or complete replacement of the prosthetic, atrophy of the urethra that would require further therapy with bulking agents or replacement of the AUS-800 or placement of an additional cuff to convert to a double cuffed system, acute and chronic infection that may require removal of the device, erosion of device parts through skin or urethra requiring removal, chronic epididymal or testicular pain, , numbness or hyperasthetic scrotal/perineal skin, the procedure uses a foreign body material (silicone polymer) that can be infected in the future, will require prophylactic antibiotics before other surgical and dental procedures to avoid possible seeding of the implant with bacteria, if the mesh does get infected then long-term antibiotics or removal of the mesh may be necessary.
You understand the procedure, general and specific risks as discussed and agree to proceed with the procedure. You also understand that not every possible complication can be listed in this counseling note and additional risks are possible, although unlikely.
To view the actual printable form for this surgery, click here: Counseling Note for Artifical Urinary Sphincter (AUS-800). To print the document, simply select print after you have opened the page. You can use that copy to sign before your surgery.
Terminology tip: If you come across words you don't understand, look them up in the On-Line Medical Dictionary.
Indications: Patient is a male with Stress urinary Incontinence.Sample Procedure Dictation:
The patient was given spinal/general anesthesia, placed in the Lithotomy position and then prepped and draped in the usual standard sterile manner. A 16 Fr Foley catheter was placed. A midline perineal incision from just below the scrotum to just above the anus was made through the skin. The subcutaneous tissues were sequentially dissected with bovie coagulation until the bulbocavernosus muscle and the Corpus Spongiosum was noted. The bulbocavernosus muscle was divided in the midline to expose the spongiosum. Lateral dissection to isolate the spongiosum was accomplished without difficulty. The location of the urethra was noted by palpating the catheter. Posterior dissection was also performed to fully isolate the spongiosum and urethra. This was then secured with a � inch penrose drain. Simultaneously a small midline incision was placed just above the symphysis pubis, dissection to the rectus fascia was accomplished, and a vertical incision was made for placement of the AUS-800 reservoir. A sub-rectus pouch was made to accommodate the reservoir and approximately 23cc of fluid. Pre-placed 2-0 interrupted vicryl sutures were placed to avoid inadvertent damage to the filled reservoir later in the case. A subcutaneous tunnel was made from the suprapubic area into the scrotum for placement of the AUS-800 scrotal pump. A similar subcutaneous tunnel was made to the approximate location of the urethral cuff. The urethral diameter was carefully measured for correct sizing of the cuff. A 4.5 cm urethral cuff size was noted. The cuff, pump, 60-70 cm reservoir and the accessory kit were all soaked in antibiotic solution until assembled. An approximate 10% Hypaque solution was formulated for instillation into the AUS-800. The entire balloon reservoir was flushed of air and 22cc of the 10% Hypaque solution was placed after the reservoir was placed in the sub-rectus pouch. The anterior abdominal wall was palpated and the expanded reservoir could not be easily identified. The pump was flushed of all large air with normal saline solution. The ends of the tubing were clamped with mosquitoes with rubber shods. A similar maneuver was accomplished with the urethral cuff. All Air and fluid was removed from the cuff and then placed around the urethra. It was noted to be of correct size. It was tested with 2.0-2.5 cc of normal saline for fit. The patients Foley catheter was removed after the bladder was filled with 250 cc of saline. Direct pressure was applied to the bladder and no urinary leakage was noted with the cuff inflated. Brisk leakage was noted with the cuff deflated. After placement of the cuff, the tubing was brought through the previously made subcutaneous tunnel to the suprapubic incision site. The pump was placed into the scrotum and loosely sutured to the most dependent portion of dartos fascia with 2- vicryl suture. The pump was palpated through the scrotal skin and noted to be in a dependent position easily pulled down for manual use. The tubing was then connected with the provided tubing connectors in a manner to leave the least amount of tubing in place and to remove all air from the system. Once fully connected the prosthesis was cycle through multiple trials of use and noted to be in good working order. The cuff was emptied of 2/3 of its fluid and the AUS-800 was then deactivated. Pressure to the bladder produced urinary leakage once the AUS was deactivated. The rectus fascia was closed with the pre-placed sutures. After copious irrigation with antibiotic solution, the Scarpa�s layer was closed with interrupted 2-0 vicryl sutures and the skin was closed with a subcuticular layer of 4-0 monocryl suture. The wound was steri-striped and bandaged. All bleeders were bovie coagulated and the wound was irrigated and inspected for further bleeding. The perineal incision was closed in three layers with 2-0 vicryl sutures. The wound was copiously irrigated with normal saline prior to subcuticular skin closure with 4-0 monocryl. The wound was bandaged. The perineal bandage was secured with an athletic supporter. A 16 French Foley catheter was placed, put to gravity drainage and secured. Patient was then awaken from anesthesia without complications and transferred to the Recovery Room (RR). The patient arrived to the RR in stable condition and without complications.
To the family and friends of patients undergoing surgery.
SCHEDULED STARTING TIME OF SURGERY:ESTIMATED LENGTH OF SURGERY:
You should plan to check in at the waiting area information desk as soon as your family member or friend has left for the Operating Room. This is the only way we can talk to you afterwards, or on occasion; reach you to give you updates on the operation's progress. If the surgery is scheduled for many hours, you can leave to eat or do other things, but you should let the information desk know that you are going to leave the area, where you are going, and how long you might be gone so that we might reach you if need be. You should be in the area before the elected time of the end of the operation.
The information deck will overhead page you or the "family of" when they receive the recovery call to let you know that the surgery has been completed. The overhead page system works ONLY on the Surgical Waiting Area and not throughout the hospital or the cafeteria.
We will plan to see you in the surgical waiting area after we have safely completed the early phases of the post-anesthesia recovery in the "Recovery Room" or PAR (Post Anesthesia Recovery). This may take up to an hour after the initial call. Sometimes, especially if another case is ready to start, we will call and talk to you. If for some reason, we have not come or called within 30 minutes, please ask the information desk to page us.
Your family member will be in the Recovery Room for 1-2 hours. This is standard recovery time, although the times vary with each individual. For example, spinal anesthetics take longer to "wear off," local anesthetics are much shorter acting. Under no circumstances are family members or friends allowed in the recovery room. The information deck will inform you of the patient's return to the room as soon as they receive the information that the patient has left recovery. At that time, they will give you the room number and direct you to the correct wing and floor.
Activity
Diet
Medication
Catheter and Wound Care
Bowel Movements
When to Contact your Doctor
Contacting Your Physician
Dr. Bauer can be contacted by calling the number listed at the top of the page. You may also call the hospital to have them contact us. Please do not hesitate to call with any questions or concerns.
Frequently Asked Questions after surgery
This section is under construction.